This week, a piece of news about cancer has found its way into the dense web of current political, economic and military information: an international consortium led by the Barcelona Institute for Biomedical Research (IRB) has just completed the pre-clinical studies that place the MCLA-158 antibody at the head of the race to find the first drug to target cancer stem cells from solid tumors.
Cancer stem cells? Until recently, we believed that cancer was essentially a homogeneous mass of rapidly proliferating cells. For this reason, the therapies that were emerging focused on that: on eliminating those “highly proliferative” cells. In recent decades, we have learned that there is enormous diversity in cancer cells. Both in proliferation and differentiation.
What’s more, in recent years, we discovered cancer stem cells. A small subset of cells that, as in the case of normal stem cells, “have the capacity to renew and generate the variety of proliferating and differentiated cells that make up the bulk of the tumor.” The problem is that they are not affected by the therapies we used. To put it metaphorically, we were attacking the soldiers, but the factory was still running.

Chaoyue Su et all (2021)
How to differentiate a normal stem cell from a cancerous one? Every time we discovered this problem, researchers have focused on finding mechanisms that allow us to attack some cells without compromising the rest. In this case, MCLA-158 is a bispecific antibody that recognizes two characteristic proteins of cancer stem cells (EGFR and LGR5). The idea of the team led by Eduard Batlle is that, precisely for this reason, “it should not interfere with the functioning of the body’s healthy stem cells, which are essential for the proper functioning of tissues”.

Battle, 2022
Still far from the clinic. This is a very important discovery and there are indications that the data is strong, but we need to temper our enthusiasm. These are preclinical data. We have known for a long time that only 5% of all drugs that have demonstrated their effectiveness in preclinical phases reach the market. As I say, this is not an argument against this antibody; it is rather an argument against overly sensational narratives.
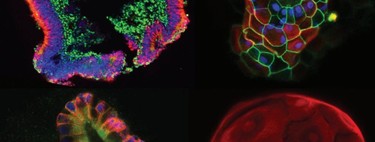
welcome organoids. However, there is something for which this work is interesting beyond what happens in the future: the use of organoids. The researchers built “a biobank that features organoids derived from colon cancer patients, organoids from colon cancer metastases in the liver, and organoids from normal, noncancerous tissue.”
“Organoids” explained to us a few years ago Benjamin Freedman, professor of medicine at the University of Washington and an expert in kidney organoids “are collections of cells on a support, like a plate, that resemble a tissue or organ of the body” . This means that, by incorporating them in the earliest phases of drug generation, he helps “identify those that are effective for most patients or even for tumors that carry a particular mutation.” It allows us to go faster.
At least that was what we believed. Now, with the good results of this study and the development of a clear methodology for the use of organoids, we can confirm it. In this way, the work of Batlle and his team opens the door to better, faster and more efficient cancer science. Even if the antibody ultimately failed to reach hospitals, its contribution would have been enormous.
Image | RAEng
George is Digismak’s reported cum editor with 13 years of experience in Journalism